Phenylketonuria (PKU) is a genetic metabolic disorder caused by a deficiency or dysfunction of the enzyme phenylalanine hydroxylase (PAH), which is primarily responsible for converting the amino acid phenylalanine (Phe) into tyrosine in the liver. Without functional PAH, phenylalanine accumulates to toxic by products of phenylalanine metabolism in the blood and brain. This buildup disrupts normal brain development and function, potentially leading to severe neurological complications including intellectual disability, behavioral issues, and seizures if left untreated. The enzyme-based understanding of PKU explains why early detection through neonatal screening and immediate dietary intervention (a low-phenylalanine diet) are critical: by limiting phenylalanine intake, the body avoids overwhelming the impaired enzymatic pathway, thereby preventing neurotoxicity.
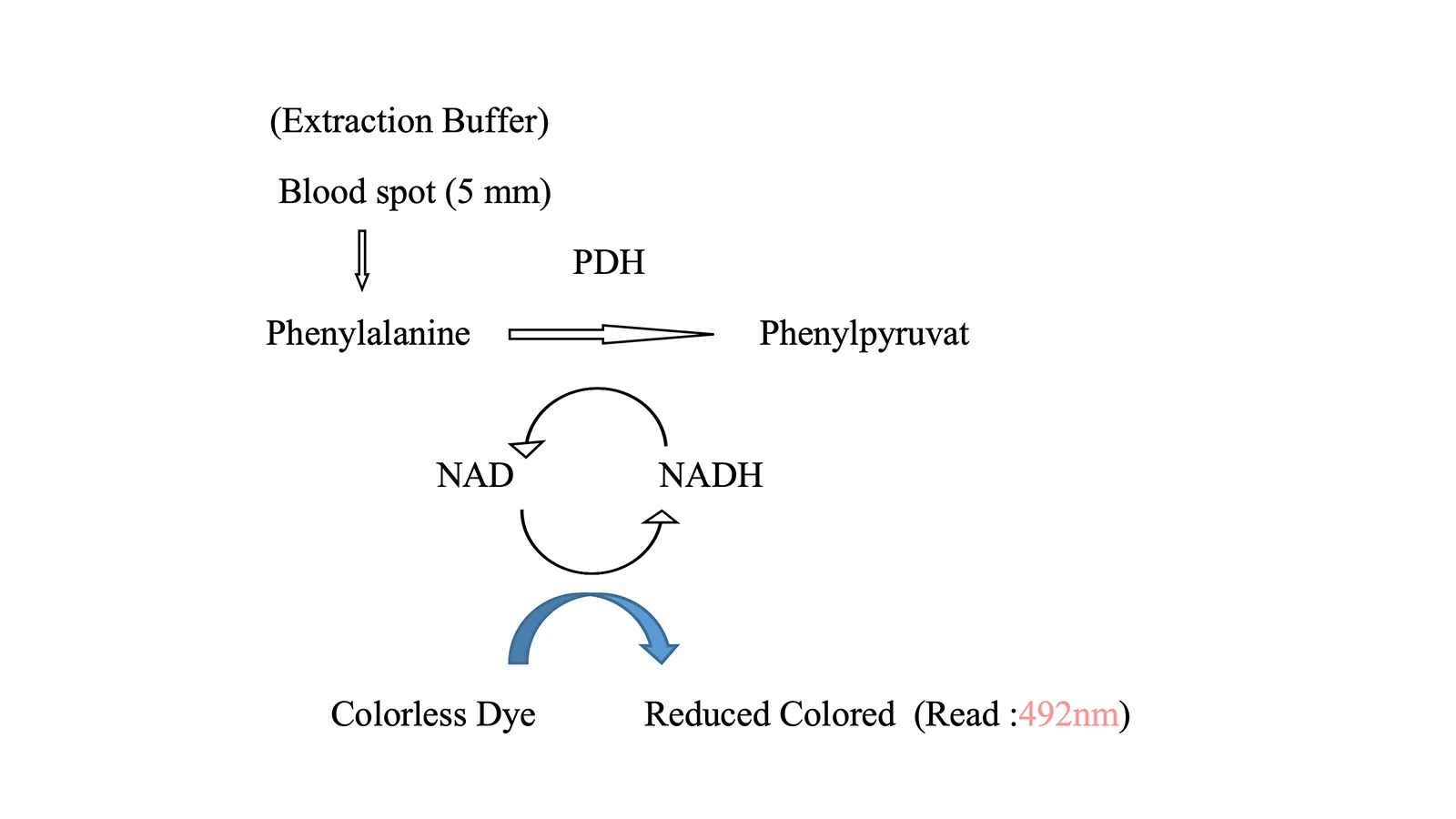
The neonatal PKU test is a routine newborn screening that detects phenylketonuria (PKU).
The test uses a heel-prick blood sample taken 24-48 hours after birth. Early detection allows immediate treatment with a low-phenylalanine diet, preventing serious complications and enabling normal development.
In 2009, Misaq Talashgaran Company, driven by its dedicated scientific team, launched the Neo-PKU newborn screening kit a milestone in domestic diagnostic innovation.
The Neo-PKU kit has since earned a distinguished position among both local and international PKU screening solutions, thanks to two key advantages:
Ready-to-use reagents eliminating the need for time-consuming preparation or dilution.
Exceptional color stability (OD) ensuring reliable results even if the final reading is delayed.
These features have made Neo-PKU the smart choice for laboratory professionals seeking fast, accurate, and worry-free phenylketonuria screening for newborns.